In an environment where much of the biotech universe is still anchored in preclinical phases or with little commercial visibility, Rocket Pharmaceuticals ($RCKT) is positioned at a critical transition phase towards commercialization in gene therapy. The proximity of the PDUFA in March 2026 for KRESLADI™., together with clinical data consisting of LAD-I and the advancement of its cardiovascular platform (Danon, ACM, BAG3), place the company at a point where the clinical evidence begins to materialize into regulatory milestones and potential revenue generation.
Before going into detail, we present a table with the current price and different valuation scenarios, defined by me, Diego García del Río, and based on the evolution of the main clinical and regulatory catalysts, including KRESLADI approval, phase 2 data at Danon, progress on ACM/BAG3, financial stability (runway until Q2 2027) and the potential for strategic agreements in cardiovascular gene therapy.
| $RCKT | CURRENT | PESSIMISTIC | NORMAL | OPTIMIST |
| PRICE (USD) | 4,69 | 2,00-3,00 | 8,00-12,00 | 12,00-18,00 |
| UPSIDE/DOWNSIDE | - | -36% to -57% | +71% to +156% | +156% to +284% |
*The target prices of Rocket Pharmaceuticals ($RCKT) mainly rely on the approval and commercial launch of the KRESLADI™ (RP-L201) in LAD-I, together with the clinical evolution of the RP-A501 in Danon disease and the regulatory progress of RP-A601 in PKP2-ACM.
The optimistic scenario assumes frictionless regulatory approval, positive data at Danon that reinforces the accelerated approval pathway and clear progress toward pivotal studies in the cardiovascular pipeline, as well as possible strategic agreements or monetization of the Priority Review Voucher (~150 M USD).
Rocket Pharmaceuticals Inc ($RCKT)
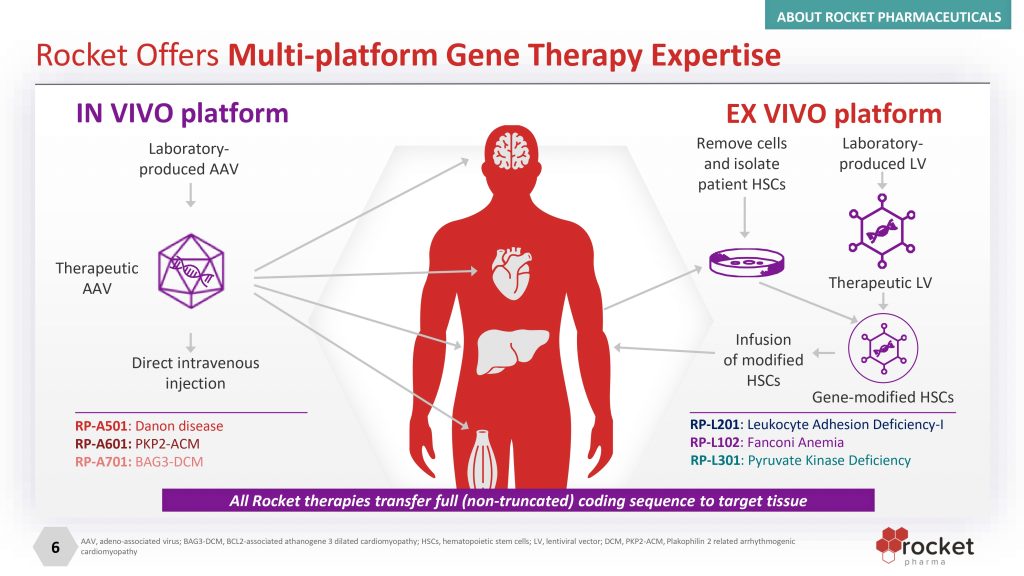
Rocket Pharmaceuticals is a biotechnology company focused on the development of gene therapies for rare diseases of genetic origin. Its approach is based on the design of treatments with single management potential, aimed at correcting underlying genetic alterations.
The company employs viral vectors as a delivery system to introduce functional copies of genes into the patient's cells, with the aim of addressing the cause of the disease instead of being limited to symptomatic treatment.
In a simple way, look for fix diseases caused by errors in the genes by introducing a healthy version of the gene into the patient's body. To do this, it uses modified viruses as “vehicles” that transport this genetic information into the cells. The idea is correcting the problem at source, instead of treating only the symptoms.
Founded in 2015, Rocket Pharmaceuticals is an integrated biotechnology company developing gene therapies through two main platforms, lentiviral vectors (LV) ex vivo for hematological disorders and adeno-associated vectors (AAV) in vivo for genetic cardiomyopathies.
This approach allows you to select the most suitable vector for each disease and build a diversified pipeline of therapies with disease-modifying potential. Beyond clinical development, the company has also invested in in-house manufacturing capabilities, with an installation of approximately 100,000 square meters in Cranbury (New Jersey) from which it produces clinical material and moves towards commercial supply.
Your most advanced candidate is KRESLADI™ (RP-L201; marnetegragene autotemcel), a gene therapy lentiviral ex vivo aimed at the treatment of severe Leukocyte Adhesion Deficiency-I (LAD-I), a pediatric disease characterized by recurrent life-threatening infections.
In the pivotal clinical trial, the treatment showed survival of 100% free of hematopoietic transplantation (HSCT) in treated patients, with follow-up of up to several years, together with a significant reduction in serious infections and improvements in relevant clinical parameters.
The application for authorization (BLA) is located under priority review, with a PDUFA date scheduled for March 28, 2026. If approved, it would mark the company's first commercial launch and could lead to the acquisition of a priority review pediatric voucher.
Beyond KRESLADI, the main driver of value in the medium term is RP-A501, a gene therapy under investigation aimed at the Danon's disease, a severe genetic cardiomyopathy associated with early heart failure and with limited therapeutic options, where cardiac transplantation is the main alternative in advanced stages.
In phase 1 (data published in NEJM 2025), the treatment has shown sustained expression of LAMP2 in cardiomyocytes for several years, together with relevant reductions in the left ventricular mass, The results showed improvements in cardiac biomarkers (BNP, cTnI) and quality of life. Likewise, the treated patients have remained alive and without the need for transplantation during the available follow-up.
The pivotal phase 2 trial is ongoing, with dosing of new cohorts planned for 2026, incorporating a optimized immunosuppressive regimen and representative material of commercial production manufactured in-house.
The cardiovascular pipeline continues to advance, with several programs in clinical development. RP-A601, directed to PKP2-arrhythmogenic cardiomyopathy (ACM), has shown in phase 1 signs of reduction of arrhythmic load, stabilization of right ventricular function and improvements in functional variables such as NYHA y KCCQ-12. The company is actively interacting with the FDA to define the pivotal study design.
On the other hand, RP-A701, focused on BAG3-dilated cardiomyopathy, The company has IND authorization and is in the preparation phase for the initiation of clinical studies in humans.
And in the hematology area, the company has prioritized its resources strategically, discontinuing the development of RP-L102 in Fanconi Anemia, while maintaining other ongoing programs, such as the one aimed at Pyruvate Kinase Deficiency.
All this shows a diversified pipeline in gene therapies, with exposure to multiple rare indications and a focus on diseases with high unmet medical need. The combination of late-stage programs with assets in early development allows for balancing the clinical risk profile and generate several turning points in the medium term.
The proximity of regulatory milestones, such as the decision on the KRESLADI, and the evolution of cardiovascular programs, in particular RP-A501, will act as the main catalyst for the validation of the technological platform and clinical and commercial execution, with a risk-return profile strongly conditioned by such events.
Rocket Pharmaceuticals is positioned within the gene therapy segment, where differentiation will depend on the consistency of clinical datathe manufacturing scalability and the ability to translate innovation into regulatory approvals and market adoption.
Beyond the current programs, the main driver of value in the medium term is RP-A501, a gene therapy under investigation aimed at the Danon's disease, a severe genetic cardiomyopathy associated with early heart failure and with limited therapeutic options, where cardiac transplantation is the main alternative in advanced stages.
In phase 1 (data published in NEJM 2025), showed long-lasting LAMP2 expression in cardiomyocytes (up to 60 months), sustained reductions in left ventricular mass (up to -48% in some patients), improvements in biomarkers (BNP, cTnI) and quality of life, with all patients alive and free of transplantation.
The pivotal phase 2 trial is ongoing, with cohort 4 dosing (3.8 × 10¹³ GC/kg) planned for the first half of 2026, using a optimized immunosuppressive regimen and commercial material manufactured in-house.
The pivotal Phase 2 trial is ongoing, with dosing of new cohorts scheduled for the first half of 2026, incorporating a optimized immunosuppressive regimen and representative material of commercial production manufactured in-house.
The most compelling thing about Rocket is that it is not pursuing the oncology hype nor the CAR-T cells massive. It is building potentially definitive cures for ultra-rare diseaseswith premium pricing, integrated manufacturing and a disciplined execution.
In a universe where many gene therapies are still struggling to come to market, Rocket has a program with PDUFA imminent, two in phase pivotal/regulatory and a dual platform which gives it a differential flexibility.
High risk, as in any other program of late stage gene therapy. Even so, the combination of survival of 100% in LAD-I, evidence of sustained protein expression y ventricular mass reduction in Danon, with a PDUFA imminent and exposure to high value markets, This helps explain the growing interest from specialized investors.
In the U.S. and Europe alone, genetic cardiomyopathies, such as Danon, PKP2-ACM y BAG3-DCM are more than 100,000 patients with critical medical need. Under typical gene therapy pricing assumptions (hundreds of thousands of USD per treatment), the addressable market exceeds ~1,000 M USD per year in a maturity scenario.
$RCKT - Fundamental Analysis: Cardiovascular and Hematology Gene Therapy, First Commercial Launch and Runway to 2027
Rocket Pharmaceuticals is at the most interesting point of its trajectory, being a fully integrated gene therapy biotech that is no longer just “promising”, but is now weeks away from its first commercial launch.
With KRESLADI™ (RP-L201) under priority review (PDUFA on March 28, 2026) and phase 1/2 data that showed survival of the 100% at LAD-I, the company goes from burn box to prepare to generate their own first recurring revenues, as it moves forward with strength in its AAV cardiovascular platform.
The company is listed on Nasdaq with a market capitalization of ~508 M USD, which places it as a small-cap within the gene therapy universe. It is slightly above the average of its peers. comparable peers (~469 M USD) and well below the general sector (~81,724 M USD), although this comparison may be misleading, since their pipeline is more advanced than that of most companies of similar size and has a in-house manufacturing capacity (~100,000 sq. ft.) already operational.
The beta of 0.45 is particularly relevant, as it reflects a lower volatility market and sector (0.64), a behavior that is characteristic of companies whose value is more closely linked to specific regulatory milestones than to the general economic cycle.
From the valuation point of view, RCKT quotes with a PER of -2.3x (TTM losses), a figure that in another context would be negative, but which here mainly reflects its pre-commercial phase. It ranks at “better” (less negative) levels than many peers (-3,4x) and below the sector (-0,5x).
The PEG of -0.09 is not yet representative, given that the company has not yet started generating revenues, although analysts project a strong growth once they materialize.
The P/B of 1.8x stands out as particularly attractive against peers (6,2x) and sector (2,6x), reflecting the value of its intellectual propertythe Cranbury GMP plant and the clinical data generated.
The P/S is technically N/A, but the implicit upside analysts estimate is relevant, we are talking about a +92,3% average, with target prices incorporating the potential of the first commercial launch and the cardiovascular milestones foreseen for 2026.
Operationally, the numbers reflect the typical profile of a biotech in the start-up phase. intensive investment, income practically non-existent, with operating loss of -228.5 M USD y net loss of -223.1 M USD in the last twelve months.
Compared to some peers that already generate revenues via partnerships, RCKT may appear to be in a worse position, although this reading is incomplete since the company is allocating capital in a disciplined way to programs that have already generated solid clinical data (survival of the 100% in LAD-I, lasting expression of LAMP2 and reduction of ventricular mass in Danon, as well as stabilization of arrhythmias in PKP2-ACM).
In terms of monetization, the projections following the possible approval of the KRESLADI aim to potential peak sales of several hundred million USD per year, in ultra-rare indications characterized by premium pricing.
The balance is undoubtedly one of the most attractive points at the moment. The company has total assets of USD 330.4 M, including 188.9 M USD in cash (as of December 31, 2025), which provides a solid runway until the second quarter of 2027.
The total debt amounts to only USD 24.9 million., The company's performance was well below that of its peers and the sector, with manageable covenants and without immediate liquidity pressure. This aspect is particularly relevant in the context of gene therapy, where many companies have recurrent recourse to dilutive capital increases.
In terms of profitability, the traditional ratios show negative figures, ROA of -52.0%, ROE of -60.3% y ROIC of -49.1%. Even so, in the context of a pre-commercial biotech in gene therapy, These metrics do not adequately reflect the underlying economic value.
The cash burn is clearly oriented to high-value catalystsas the KRESLADI PDUFA, development in phase 2 in Danon and the programs in phase 1 in ACM and BAG3, rather than exploratory initiatives without clinical visibility.
In terms of commercialization, the model has the potential to generate attractive margins, supported by therapies of single administration, with scalability, internally controlled manufacturing costs and a premium pricing characteristic of curative gene therapies.
As for the short interest, the current data show a short float of 14,55%with 14.69 million short shares out of a total of 100.98 M shares outstanding (float). The short ratio of 5.22 days implies that, at the average trading rate, it would take more than five days to cover open short positions.
This level of positioning reflects a relevant presence of bearish positions, The market, although not dominant, and leaves room for accelerated movements in the event of positive catalysts, especially around regulatory and clinical milestones.
For an investor with long time horizon and risk tolerance, this type of profile represents an opportunity aligned with phases previous to value creation events. The company combines a first launch imminent, a consolidated dual technology platform and a financial runway which extends until the expected start of revenue generation.
In an environment where many biotechs do not reach the commercial stage, Rocket Pharmaceuticals shows, for the time being, a consistent execution and a clearly defined roadmap for the period from the 2026-2028.

$RCKT - Clinical Roadmap, Value Catalysts and Potential Revaluation
The main catalysts that may mark the valuation trajectory of Rocket Pharmaceuticals are as follows, especially following the updates to the Corporate Overview (February 2026) and the fourth quarter 2025 financial results, where the company has reaffirmed its priority focus on the AAV cardiovascular platform and the imminent commercial launch of KRESLADI™.:
- March 28, 2026 - PDUFA for KRESLADI™ (RP-L201) in severe LAD-I.
The FDA has established the target decision date for the re-submitted BLA of marnetegragene autotemcel in Leukocyte Adhesion Deficiency-I (LAD-I). Phase 1/2 data have shown survival of 100% at 12 months, The company's primary and secondary endpoint compliance and a favorable tolerability profile.
An eventual approval would mark Rocket's first commercial launch, enable it to obtain a Priority Review Voucher (~150 M USD estimated) and validate its lentiviral platform ex vivo.
This binary catalyst could have a significant impact on the share price, with a possible inflow of institutional flows and a re-rating of multiples in a favorable scenario.
- H1 2026 - Resumption and dosing of cohort 4 in the pivotal phase 2 trial of RP-A501 in Danon's disease.
After the lifting of the clinical hold in August 2025 (in less than three months), Rocket has resumed dosing in pediatric and adult/adolescent patients with a optimized immunosuppressive regimen (3.8 × 10¹³ GC/kg).
Phase 1 data already showed durable LAMP2 expression, sustained reductions in left ventricular mass (up to -48%) and improvements in biomarkers and quality of life.
The publication of new positive clinical data would reinforce the accelerated approval thesis and could translate into a relevant impact on titration, especially if stabilization of cardiac function is confirmed.
- Mid-2026 - First Phase 1 patient dosing of RP-A701 in BAG3-Dilated Cardiomyopathy
Start of the first human study for this candidate AAV in genetic dilated cardiomyopathy. With the IND already authorized and regulatory alignment, this milestone validates the expansion of the cardiovascular platform and could attract greater attention from KOLs and potential strategic partners.
In a favorable scenario, it could generate a positive impact on valuation, especially due to its early stage pipeline expansion nature.
- 2026 (ongoing) - Advancement with FDA for pivotal design of RP-A601 in PKP2-Arrhythmogenic Cardiomyopathy
Rocket maintains active interaction with FDA following preliminary phase 1 data (presented at ASGCT 2025 and subsequently updated), which showed PKP2 expression, stabilization of arrhythmias, improvement in right ventricular function and quality of life. Phase 1 recruitment is still ongoing.
Regulatory alignment for a potential pivotal Phase 2 study would be a relevant catalyst, reducing regulatory uncertainty and providing greater visibility on the clinical development pathway.
- Potential for strategic partnerships, pipeline expansion or M&A in cardiovascular gene therapy
With KRESLADI regulatory decision, solid data on Danon and ACM, and a own GMP plant (~100,000 sq. ft.), Rocket is positioned as a potentially attractive platform for big pharma with an interest in genetic cardiology. An agreement of licensing, collaboration or even a corporate operation (with a current capitalization of ~500 M USD) would constitute a transformational catalyst, albeit of a speculative nature and conditional on regulatory and clinical execution. In any case, it could reinforce the financial runway and accelerate geographic expansion.
Rocket does not rely on a single binary event: it combines an PDUFA imminent, programs in advanced stages and a dual platform (LV + AAV) with in-house manufacturing that helps mitigate operational risks. The milestones foreseen between March and June 2026 will be decisive for the transition from a late-stage biotech towards a company with a commercial profile in gene therapy.
Revaluation Scenarios
In a upward scenario, driven by the approval of KRESLADI™ in the March 28, 2026 PDUFA (with the entire Phase 1/2 data package fulfilled and no material deficiencies), positive data from Cohort 4 in the pivotal Phase 2 trial of RP-A501 in Danon Disease during H1 2026, and clear progress toward the pivotal design of RP-A601 in PKP2-ACM, the target price could be in the range of $12-18which would imply a upside from +140% to +260% from current levels (~1TP4Q5). This scenario assumes extended runway with no relevant dilution, possible Priority Review Voucher (~150 M USD) and first commercial revenues in 2026, expanding the market capitalization to 1,200-1,800 M USD, This is in line with other gene therapy companies that have achieved their first launch (such as Bluebird or Sarepta in their initial stages of commercialization).
In a base scenario, With execution aligned to the February 2026 corporate calendar (KRESLADI approval in March, uneventful resumption of dosing at Danon and regulatory progress on ACM and BAG3), with no major surprises but with consistent safety and biomarker data, the target price would be around $8-12, equivalent to a revaluation from +60% to +140%. This case contemplates a controlled burn, constructive analytical consensus (average targets ~$13-14) and P/B multiples in the 4-6x range after the first launch, leading the capitalization to 800-1,200 M USD in a moderately favorable biotech environment, with focus on transition to commercial enterprise and scalability of its ~100,000 sq. ft. GMP facility.
In a bearish scenario, conditional on a delay or Complete Response Letter in the KRESLADI PDUFA, safety or clinical hold issues in the Danon pediatric cohort, or manufacturing difficulties that reduce the runway before mid-2027, coupled with an adverse biotech environment (high rates or funding constraint), the price could be adjusted towards $2-3, which would represent a downside risk from -40% to -60%. This scenario would include possible additional dilution (~20-30% via ATM or equity) to bolster cash, deterioration of investor sentiment and short squeeze, contracting the capitalization to 200-300 M USD and shifting key milestones to 2028, reflecting the inherent volatility of gene therapy companies in commercial transition.
$RCKT - Technical Analysis: Technical Consolidation vs. Catalyst Context
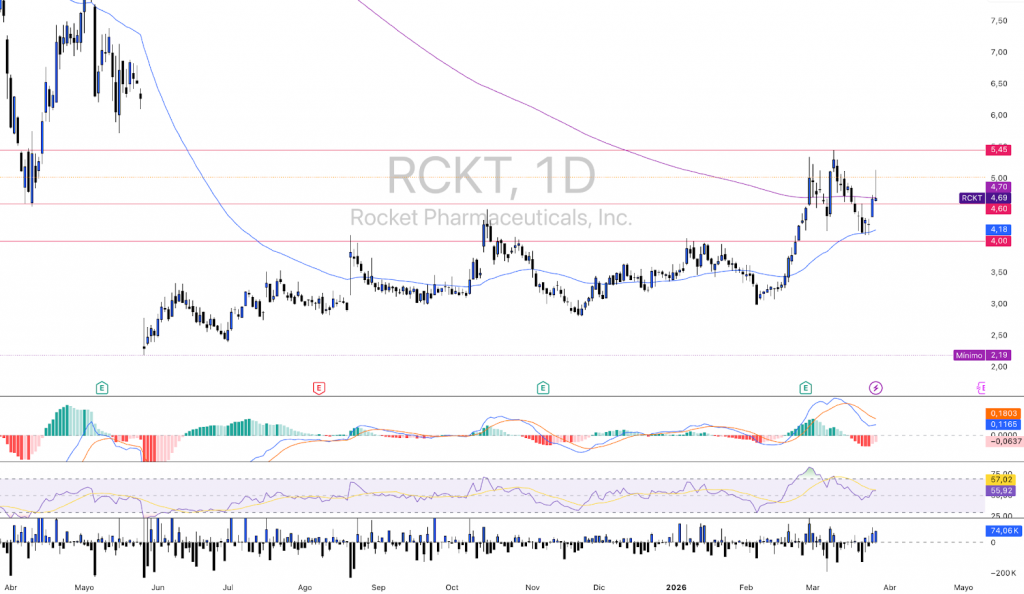
After a downward gap and a prolonged period of consolidation during much of the previous year, $RCKT began a significant upward momentum in early 2026, breaking through key structural resistance. From its recent highs (in the area of the $5,40-$5,45), the stock has entered a natural corrective phase, retreating back towards the $4.60-$4.70. This behavior is common after aggressive breakouts, where part of the market takes profits, testing the strength of the new support levels.
The daily chart reflects a clear transition from a prolonged sideways range to an uptrend structure. rising minima in the medium term. However, the downward crossover on recent closes introduces a short-term weakness bias. The price seems to be consolidating towards the former resistance zone, now turned into theoretical support, a necessary move to validate the underlying trend change.
At current levels (~$4.60-$4.70), the stock is testing a relevant intermediate support zone, with key lower support at $4,15-$4,20 and critical level in $4,00. The defense of this range will be decisive: the appearance of reversal candles would suggest supply absorption by strong hands, while a sustained loss could return the price to the previous consolidation range.
The first technical resistance zone is located in the area of the $5,00, The price is trading at the recent relative highs. A breakout with volume would open the door to a new test of the $5,20, with potential extension to higher levels in case the momentum is confirmed.
The RSI (14) is located in 44,71, after normalizing the overbought readings registered during the previous impulse. This behavior indicates a healthy “cooling” of the price and leaves room for further upside. In biotech assets, RSI stabilization in the 40-50 range is usually consistent with consolidating uptrends.
For its part, the MACD confirms the current corrective phase: the line has crossed its signal to the downside and the histogram remains in negative territory (-0.0637), reflecting a short-term bearish momentum. This context suggests caution until signs of exhaustion in the selling pressure are observed.
At current levels, and provided that the support from $4,00-$4,15 the asset is maintained, the asset presents an interesting structure in terms of risk/return asymmetry for strategies for swing trading. The key will be the confirmation of demand in this area, which would validate the continuity of the bullish scenario in the medium term.
Synthesis
Rocket Pharmaceuticals is not just another experimental-stage biotech, it is a company that is crossing the most complex threshold in the industry, moving from clinical promise to commercial reality in gene therapy. I am not talking about incremental optimizations, but about interventions with curative potential in ultra-rare diseases, where single administration can completely redefine the therapeutic standard. His approach, based on viral vectors (lentivirals ex vivo and AAV in vivo), seeks to directly correcting the genetic defect, attacking the root of the problem instead of mitigating symptoms.
As of March 2026, with a capitalization of ~508 M USD, RCKT sits somewhere in between undervalued small-cap and late-stage biotech in transition. It trades with a PER of -2.3x (typical in the pre-revenue phase), but with a P/B of 1.8x which is particularly attractive compared to comparables (~6.2x), reflecting a market that has not yet fully discounted the value of its IP, its proprietary GMP plant and, above all, the imminent monetization via KRESLADI™.. The implicit consensus points to an upside of approx. 90%, although more aggressive scenarios, linked to clinical and regulatory execution, raise that range significantly.
Financially, the company presents a surprisingly solid structure for its stage. With ~188.9 M USD in cash and a contained debt (~24.9 M USD), the runway extends to Q2 2027, This allows it to meet key milestones without resorting to aggressive dilution, one of the main structural risks in the sector. The burn is clearly focused on high impact catalysts (PDUFA, pivotal trials in Danon, cardiovascular expansion), not to early exploration without visibility.
The real turning point is the March 28, 2026 PDUFA for KRESLADI™ at LAD-I.. It is not just a binary event, it is the validation of the entire lentiviral platform ex vivo. If approved, Rocket moves from consuming capital to generating revenue in a niche with premium pricing, in addition to potentially capturing a Priority Review Voucher (~150 M USD), This would have a direct impact on the balance sheet and institutional perception. At the same time, RP-A501 in Danon disease has already shown high quality clinical data (sustained expression of LAMP2, reduction of ventricular mass, transplant-free survival), positioning itself as the true medium-term value driver within the cardiovascular platform.
Technically, it is in a phase of consolidation after momentum, trading at around $4.60-$4.70, with critical support in the area $4,00-$4,15. The RSI (~45) reflects neutrality after overbought unloading, while the negative MACD indicates short-term bearish pressure, typical before catalytic events. The short interest (~14.5%) not extreme, but sufficient to amplify movements if fundamental validation occurs. The beta of 0.45 reinforces the idea that their behavior is more linked to idiosyncratic events than to the general market.
The catalysts are perfectly defined in calendar, PDUFA in March (key event), Phase 2 data on Danon in H1 2026, regulatory progress on ACM (RP-A601), clinical initiation on BAG3 (RP-A701) and potential strategic moves (partnerships or M&A). It does not depend on a single asset, but rather on a platform with multiple avenues of value creation.
RCKT is not a “lottery ticket”, but neither is it a defensive profile, although it is normal, due to the nature of this type of company. transition thesis, The risk is clear and binary (a setback in PDUFA could lead to $2-3, -40%/-60%). The risk is clear and binary (a setback in the PDUFA could take it to $2-3, -40%/-60%), but the upside is equally defined (1TP4Q8-12 base scenario, 1TP4Q12-18 bullish scenario, +70% to +250%). For an investor who understands the dynamics of gene therapies and the weight of regulatory milestones, Rocket represents that exact point where the market still hesitates and where the greatest asymmetries are built.