The area of autoimmune diseases is, together with oncology, the therapeutic area with the highest number of highest density of clinical development in biopharma at this time. It is no coincidence, these are chronic, highly prevalent pathologies, with patients requiring lifelong treatment and where the standard of care has not been substantially renewed for decades. When a genuinely differentiated drug enters this space, the market knows how to reward it., Benlysta, Skyrizi, Dupixent are examples of what happens when biology and commercial execution align. The problem is that the market also has a tendency to ignore names that have yet to hit the institutional noise, even though the data is already on the table. Aurinia Pharmaceuticals has long been in that category.
We are talking about a company that closed 2025 with $271 M in sales of its only marketed product, growing at 25% year-on-year, $136 M in operating cash flow, more than three times the previous year, $398 M in cash with no relevant debt pressure, and a program of active share buyback that has returned nearly $100 M to shareholders in twelve months. Such a company, in any other sector, would trade at a significant premium. In mid-cap biopharma, it trades at a discount because it has only one marketed product and the market does not yet know what to do with the second asset in development.
That is exactly the inefficiency that makes this analysis interesting. The concentration discount that the market applies to AUPH has partial logic; if Lupkynis disappoints, there is no net. But that same discount ignores that aritinercept (AUR200) just released Phase 1 data positioning the drug as one of the most potent dual BAFF/APRIL inhibitors in its class, in a therapeutic space, B-cell mediated autoimmune, where the potential TAM has nothing to do with that of lupus nephritis. It also ignores that the company has enough cash to fund the development of that pipeline without diluting anyone, and that the ACR 2025 guidelines have shielded Lupkynis at the forefront just as a new competitor, Roche's Gazyva, has just entered the market.
The result is a stock that trades as if the future is uncertain when the present is already resolved. That gap between what the numbers say and what the price reflects is, in my opinion, the thesis.
Before going into the detail of the analysis, I present below the different valuation scenarios I estimate for Aurinia Pharmaceuticals ($AUPH), defined based on Lupkynis' commercial evolution towards the $305-315M guidance by 2026, the clinical activation of aritinercept in new autoimmune indications in the first half, the company's use of its $398M in cash, repurchases, M&A or pipeline investment, and the impact that the entry of Roche's Gazyva (obinutuzumab) will have on prescribing patterns in lupus nephritis. These are the four axes that, in my opinion, will determine whether or not the position I took in CALL STK $20 expiring in October has real runway within the time horizon.
| $AUPH | CURRENT | PESSIMISTIC | NORMAL | OPTIMIST |
| PRICE (USD) | 16,25 | 10,00-12,00 | 20,00-25,00 | 30,00-38,00 |
| UPSIDE/DOWNSIDE | - | -26% to -38% | +23% to +54% | +85% to +134% |
Aurinia Pharmaceuticals Inc. ($AUPH)
Aurinia Pharmaceuticals is a Canadian-American biopharma with an exclusive focus on high unmet need autoimmune diseases, i.e. with unmet medical needs where current therapeutic options are limited, ineffective or non-existent. It is not an early stage company borrowing from the market for a promising pipeline, it has a marketed, FDA-approved product that is growing at double digits and generating real operating cash. That's a critical distinction in an industry where most similarly capitalized names continue to burn capital with no visibility of profitability.
Its main asset, Lupkynis (voclosporin), was approved by the FDA in January 2021 as the first oral therapy for the treatment of adults with active lupus nephritis. Not a marginal niche drug, lupus nephritis is the most serious renal complication of systemic lupus erythematosus, affecting between the 40 and 60% of patients with lupus, and its inappropriate management consistently leads to end-stage renal failure. The differentiation of voclosporin over classical calcineurin inhibitors (cyclosporine and tacrolimus) is not marketing, its more predictable pharmacokinetic profile, without the need for intensive monitoring of levels, plus the renal protection documented in the AURORA 1 and AURORA 2 studies, are real clinical arguments that nephrologists ponder when prescribing. The mechanism also includes stabilization of the renal podocyte, an additional action on classic cyclosporine, which translates into better preservation of the glomerular architecture in the long term.
The second asset under development, Aritinercept (AUR200), concentrates the potential of re-rating. It is a dual inhibitor of BAFF and APRIL designed with a differential molecular architecture: it employs a BCMA-based binding domain, with higher intrinsic affinity for APRIL vs. TACI-engineered used by other dual inhibitors, and an Fc IgG4 with minimal effector function.
BAFF and APRIL are key cytokines in the survival and differentiation of B cells and plasmablasts, responsible for the production of pathological autoantibodies in a broad spectrum of autoimmune diseases. If the clinical profile confirms the signals observed in Phase 1, the accessible TAM for Aritinercept would be orders of magnitude higher than that of Lupkynis.

Pipeline and Clinical Analysis: Lupkynis holds, aritinercept starts to talk
Lupkynis has been on the market for five years and long-term data support the commercial thesis. The AURORA study demonstrated a significantly superior complete renal response rate to placebo plus mycophenolate mofetil, with a safety profile that allowed approval without the usual restrictions for calcineurin inhibitors in transplantation. AURORA, the two-year continuation study, confirmed durability of response and sustained renal safety profile. According to the Spherix Global Insights patient chart audit published in March 2026, specifically: «Spherix Global Insights Finds Benlysta Exposure Expanding and Rheumatologists Increasingly Turning to Lupkynis in Lupus Nephritis«, approximately 1 in 5 lupus nephritis patients managed by nephrologists in the U.S. currently receives a calcineurin inhibitor, and Lupkynis clearly dominates that class.. The updated American College of Rheumatology guidelines in 2025 reinforced this positioning by conditionally recommending triple therapy (glucocorticoids, MMF or cyclophosphamide, and Benlysta or a CNI) for patients de novo, in flare, or with active class III or IV disease. That first-line recommendation is a regulatory shield that protects Lupkynis growth.
The competitive landscape in lupus nephritis became more complicated in October 2025 with the approval of Gazyva (obinutuzumab) from Roche/Genentech, the third drug approved specifically for this indication. REGENCY data are strong, 46,40% complete renal response rate vs. 33,10% placebo over standard of care. Specialists surveyed by Spherix three months after launch describe Gazyva as a favorite for efficacy in active or refractory disease. The important nuance is that Gazyva and Lupkynis are not directly competing for the same patient at the same time, Lupkynis maintains real advantages in oral access, out-of-pocket cost and absence of eGFR restrictions that do apply to Gazyva.. The real-world data show that Gazyva's direct competitive pressure falls primarily on Benlysta and off-label use of rituximab, not on front-line Lupkynis.
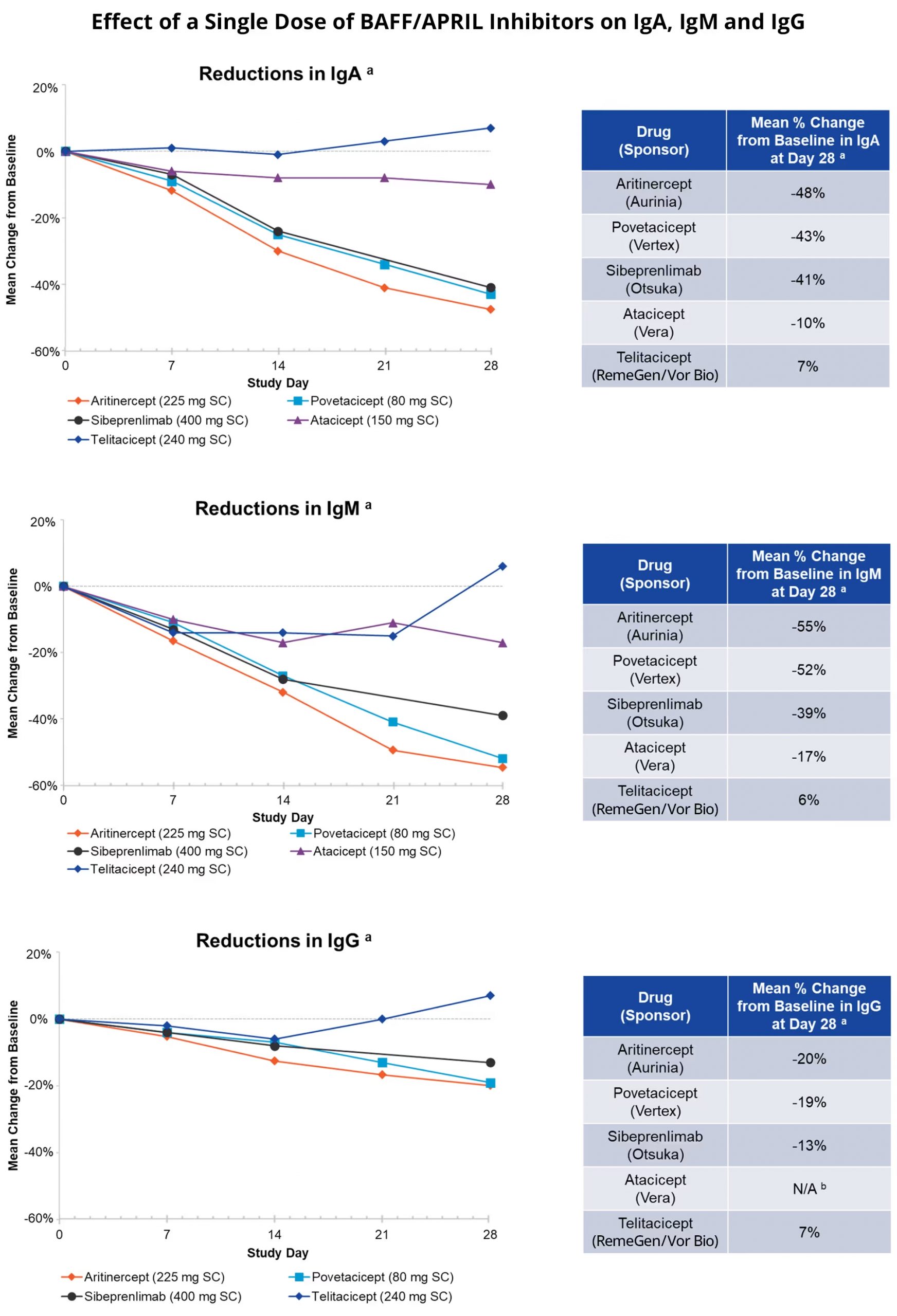
For aritinercept, the results of the Phase 1 SAD study released in June 2025 exceeded market expectations. In 61 healthy volunteers, single subcutaneous doses generated average reductions of up to 48% in IgA, 55% in IgM and 20% in IgG at day 28., without related adverse events ≥ grade 3, without SAEs and without discontinuations. The magnitude and duration of immunoglobulin suppression with single dose supports a monthly subcutaneous regimen, a relevant logistic advantage in chronic diseases where adherence is a determining factor.
What adds weight to these data is the comparison with the other BAFF/APRIL dual inhibitors in development:
- Atacicept (Vera Therapeutics)
- Povetacicept (Novartis)
- Sibeprenlimab (Otsuka)
- Telitacicept (RemeGen)
Aurinia has published SAD crossover data showing that aritinercept, at doses equivalent to those selected by competing sponsors for their Phase 3, generates equal or greater IgA and IgM reductions. The most relevant clinical precedent as a class validation signal is atacicept in IgA nephropathy, its ORIGIN study (Phase 2b, Kidney International 2024) demonstrated 25% reduction in UPCR versus placebo, with the primary endpoint met at 24 weeks. Whether aritinercept replicates that signal with greater pharmacologic potency, suggested by the Phase 1 profile, the indication could be transformational for the company.
The company has already initiated a first clinical trial in an autoimmune indication, without formally disclosing which one, and has committed to initiate a second test in the first half of 2026, according to CEO Peter Greenleaf's statements at the February earning call. The most likely candidates by mechanism, unmet need and precedent in the class are IgA nephropathy, Sjögren's syndrome and systemic SLE. IgA nephropathy affects between 130,000 and 150,000 people in the U.S. with increasing prevalence and an approved drug market that has only just opened.
$AUPH - Fundamental Analysis: Lupkynis Business Expansion, Cash Flow Generation and Pipeline Optionality
Aurinia is currently trading at around $16.25 with a market capitalization of approximately $2.14 B. With $398 M in cash, the implied enterprise value that the market assigns to the operating business plus the pipeline is approximately $1.74 B.
For a company guiding $315-325M in total revenues in 2026 with expanding operating margins, that's a multiple of 5-6x projected sales, a valuation that starts to become demanding if pipeline catalysts don't materialize, but still reasonable if aritinercept generates clinical signal in a high TAM indication.
Lupkynis' net sales amounted to $271.3 M in 2025, a growth of 25% over the previous year's $216.2 M, which in turn had grown by 36% over 2023. Quarterly progression has been consistent: Q1 2025 with $62.5 M total revenue (+24% YoY), Q2 with $70 M (+22%), Q3 with $73.5 M (+27%) and Q4 with $77.1 M (+29%), beating consensus estimates in each quarter. Aurinia reports more than 2,300 active patients in the U.S. receiving Lupkynis, with international penetration through Otsuka in Europe and Japan still at an early stage, indicating that organic growth has real scope without the need for new indications.
The Q4 2025 result showed a EPS of $1.53 vs. consensus of $0.21, 1TP4Q173m tax benefit from the release of the valuation allowance on deferred assets. Adjusting for this non-recurring impact, recurring operating EPS remains positive and growing. The 2025 operating cash flow was $135.7M, an increase of 206% over 2024., with a net margin of 29% in the fourth quarter. These are metrics of a company operating with real profitability discipline, not a biotech in cash burn mode.
The share repurchase program deserves attention, in 2025 the company repurchased 12.2 M shares per $98.2M, $150M, with the Board having approved an additional $150M. With a cash to debt ratio of 4.5x and short interest at around 8% of float, the financial structure has no pressures to justify a high risk premium. The real fundamental risk remains concentration: Lupkynis is virtually the only source of revenue, and Gazyva's entry into the market adds a competitive variable that did not exist six months ago. Any slowdown in growth will be read as a loss of share, regardless of the cause.
$AUPH - Catalysts: Visibility on Lupkynis and Binary Events on Aritinercept
The next concrete event with the ability to move the price is the Q1 2026 results release, expected April 30. The market will be looking for confirmation that Lupkynis remains on track for annual guidance of $305-315M, and that the entry of Gazyva, approved just six months ago, is not eroding prescribing dynamics. A clean quarter in line with estimates would have moderate impact on price; an upward revision to guidance or constructive comments on aritinercept could generate a 10-20% move.
The catalyst with the greatest potential for impact in the 2026 horizon is the confirmation of the initiation of the second clinical trial of aritinercept, committed for H1 2026. It is not just the start of the trial, the formal disclosure of the chosen indication is the real re-rating event. If the indication is IgA nephropathy, where atacicept has already validated the class in Phase 2b and there are two BAFF/APRIL inhibitors in advanced development, the market will have context to value a potential multi-billion dollar TAM. An indication of that magnitude could generate a move of between 25 and 45%. If preliminary data from the first trial already initiated were added to that, the potential impact would escalate to the 50-80% range.
The potential of a company event is not negligible either. With $398 M in cash on $2.14 B of capitalization, Aurinia has an attractive balance sheet for a strategic acquirer. AstraZeneca has Saphnelo approved in SLE but no presence in lupus nephritis. Roche just entered the indication with Gazyva and is building position. Bristol-Myers Squibb has autoimmune exposure but nothing specific in renal lupus. If aritinercept demonstrates signal in IgA nephropathy or Sjögren's, the asset could accelerate conversations that are not visible to the market today.
The most relevant binary risks are three:
- The first is that Gazyva wins share from Lupkynis faster than anticipatedSpherix's initial data does not show such a pattern, but the 2026 quarterly monitoring will be decisive.
- The second is any disappointment in aritinercept clinical trialsAn unexpected safety signal in Phase IIa or lack of pharmacological efficacy would suddenly eliminate the pipeline premium.
- The third, of lower probability but maximum impact, would be a adverse regulatory event related to Lupkynis, which with five years in the market and thousands of patients exposed has a low but non-zero probability.This would be a relevant catalyst given the current market capitalization (~312 M USD).
Revaluation Scenarios
In a optimistic scenario requires that two things occur in the same time horizon:
- The first: Preliminary data for aritinercept in a high TAM indication showing efficacy signal, no need for Phase 3, a biomarker readout or secondary endpoint confirming pharmacological activity in real patients is sufficient. If atacicept demonstrated -25% in UPCR in IgA nephropathy in Phase 2b, and aritinercept has better pharmacological profile in Phase 1, an equivalent or higher signal would transform the company's narrative discontinuously.
- The second: That Lupkynis does not disappoint. Lupkynis growth should remain above 15% in 2026 and guidance not be revised downward.
With both conditions fulfilled, a re-rating to 8-10x forward sales, the multiple that the market typically assigns to autoimmune platforms with two assets in the clinic, would drive the price to the $30-38 range.
A corporate event, such as a partnership or partial acquisition of aritinercept, could act as an accelerator for this scenario without the need to wait for data.
In a base scenario, does not demand miracles, it demands execution.
Lupkynis grows in line with $305-315 M guidance, quarterly data confirm that Gazyva is not eroding top line, and Aurinia announces before the close of the first half year the second aritinercept indication with formal trial initiation.
No efficacy data is needed yet, as long as the market can start sizing the TAM of IgA nephropathy or Sjögren's, the multiple expands.
In this context, a price of $20-25 would imply an EV of between $2.2 B and $2.8 B on a business that generates $136 M of OCF and grows at 25% per year, a completely justifiable valuation without the need to discount speculative scenarios.
The timing catalyst in this scenario is the Q1 2026 earnings on April 30, if Greenleaf confirms the start of the second trial on that call, the move to $20 could materialize within weeks.
In a bearish scenario does not require anything to go particularly wrong, only that the market stops giving credit.
Gazyva has been in the market for six months and if the Q2 or Q3 2026 prescription data starts to show real slowdown in Lupkynis, the market will interpret loss of share even if the cause is something else.
Downwardly revised guidance from the current $305-315 M, even marginally, would be sufficient for a 20-25% correction from these levels.
Add to that silence on aritinercept, no second indication announcement, no preliminary data, no corporate news, and the pipeline premium that is built into the price today disappears.
The relevant support in this scenario is the $12-12.93 area, where the long-term averages and the gap fill converge. Below $12 the fundamental thesis is revalued from zero.
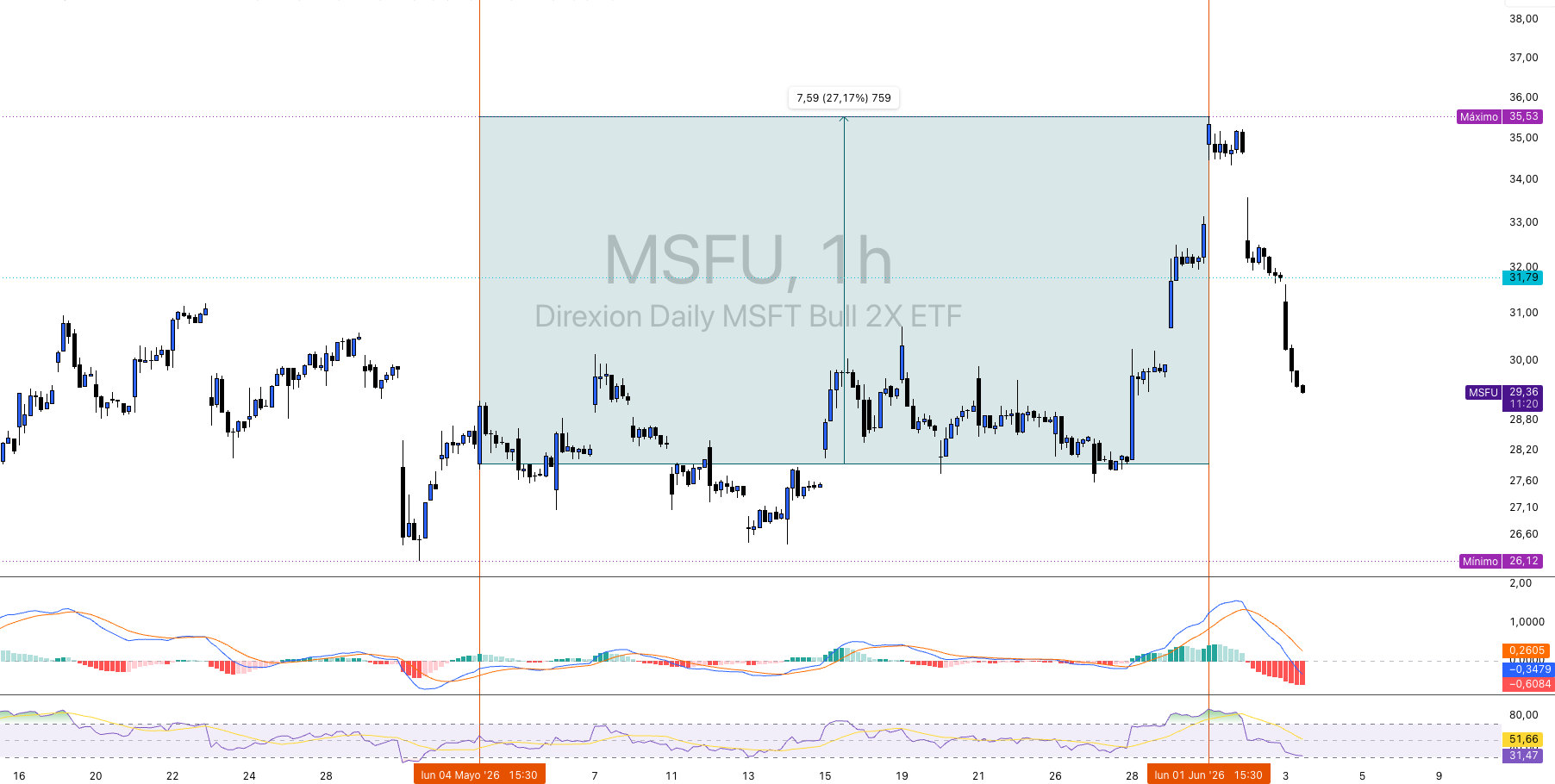
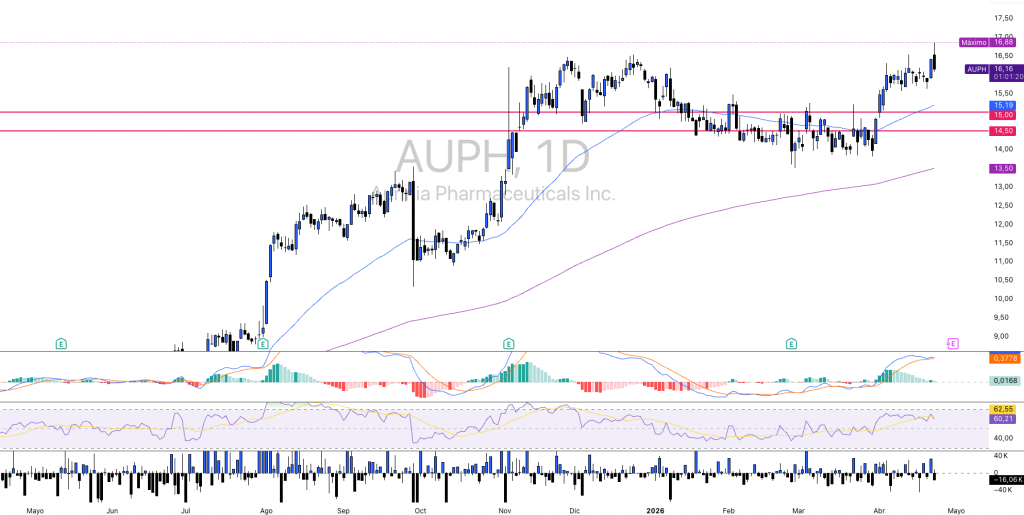
$AUPH - Technical Analysis: Range Compression and Imminent Breakout
The daily chart of AUPH shows a structure that clearly replicates the history of the stock over the past twelve months, a prolonged accumulation phase in the zone of $6-8, an impulsive move between July and October 2025 that took the price from lows of $6.55 to highs of $6.55. $16,54 (ATH), and a subsequent consolidation in the 1TP4Q14-16.50 range that has been building for several months. The current price of ~1TP4Q16.25 is at the high end of that consolidation range, just a few cents shy of the cycle's absolute high.
The immediate supports are in $14,50-15,00 being a congestion zone of the last three months, with major support at 1TP4Q12.00-12.93, which corresponds to the gap fill zone and long-term moving average reference. Key resistance is $16.42-16.54, the cycle highs that price has touched and rejected on at least two occasions. A clean break of that level with volume would confirm the bullish extension and open the way to $20-22 as the next relevant technical target.
The MACD (12,26,9) shows the fast line at 0.4013 and the signal at 0.3736, with positive but contracting histogram (0.0277) signaling bullish momentum present but decelerating. The RSI (14) stands at 65.07, above its signal average at 62.73, at upper-middle zone. It is not technically overbought, but the stock is priced where fundamental catalysts take on greater relative relevance than pure momentum to generate additional extension.
The recent volume is lower than the peaks recorded during the impulsive move of Summer-Fall 2025, consistent with a compression phase. The structure suggests that the stock is in a decision zone, either the price breaks above $16.54 with volume and confirms bullish extension, or consolidates and corrects towards $14.50-15.00 support before a new attempt. The technical bias is constructive as long as it remains above the 200-session average.
Synthesis
Aurinia Pharmaceuticals ($AUPH) is an autoimmune biopharma that I believe is in that uncomfortable spot for the market, too “real” to be treated as speculative biotech, but still without the recognition that established platforms receive. Not because it lacks growth, but because it continues to be perceived as a one-product story. And therein lies the inefficiency. We are not talking about a pre-commercial stage company; we are talking about a business that already generates relevant revenues with Lupkynis, but also has a second lever, aritinercept, which can extend the reach far beyond lupus nephritis.
Financially, AUPH does not fit the typical industry profile. With ~$271M in sales in 2025 (+25% YoY), ~$136M in operating cash flow and ~$398M in cash, the balance sheet is more typical of a profitable mid-cap than a developing biotech. At current multiples (~5-6x forward sales), the market is valuing the existing business, but still assigns little value to the pipeline. This mismatch is key, the downside is partially covered for a working business, while the upside depends on the second asset starting to “exist” in terms of market perception.
That second active is aritinercept, a dual BAFF/APRIL inhibitor in a space where there is already prior clinical validation. Here you don't need an immediate Phase 3 success to move the action; something simpler but just as powerful at the narrative level, a clear indication, an ongoing trial and early signs of drug activity in patients is enough. If that happens in areas such as IgA nephropathy or Sjögren's, the market may start to value Aurinia as a platform history, not as a single-asset play.
The main risk is not in the pipeline, but in the current core. Gazyva's entry introduces competitive pressure in lupus nephritis, and although early data suggests it does not compete directly with Lupkynis in the front line, any slowdown in prescribing is going to be interpreted as a loss of share. In a concentrated company, the narrative changes quickly if growth cools, even if fundamentals remain strong.
In terms of market structure, the chart reflects well that tension, after the rally from 1TP4Q6-8 to ~1TP4Q16, the stock has been consolidating just below highs for months. There is no clear distribution, but no confirmed breakout either. It is an area where the price stops moving by inertia and starts to depend on catalysts. Either there is a fundamental trigger and it breaks to the upside, or the price needs to correct and rebuild its base.
The next few months are fairly binary in terms of narrative, a Q1 2026 to validate that Lupkynis is still on trajectory, announcement of the second aritinercept indication in H1, and any initial clinical signals. If all of that fits, the $20-25 range in baseline scenario is reasonable without the need to assume heroics. If relevant clinical signal also appears, the market can start paying platform multiples. If not, support at ~$14.5-15 becomes the area where the thesis is called into question again.